Comparison of confirmed and probable COVID-19 patients in the intensive care unit during the normalization period
DOI:
https://doi.org/10.17305/bjbms.2021.6657Keywords:
COVID-19, SARS-CoV-2, confirmed, probable, intensive care unit, normalization periodAbstract
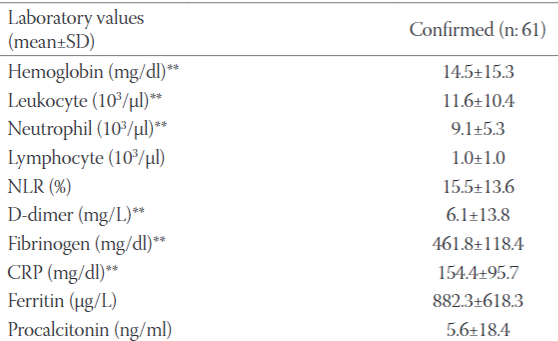
The decrease in social distance together with the normalization period as of June 1, 2020 in our country caused an increase in the number of COVID 19 patients. Our aim was to compare the demographic features, clinical courses and outcomes of confirmed and probable coronavirus disease 2019 (COVID-19) patients admitted to our intensive care unit (ICU) during the normalization period. Critically ill 128 COVID-19 patients between June 1 - December 2, 2020 were analyzed retrospectively. The mean age was 69.7±15.5y (61.7% male). Sixty-one patients (47.7%) were confirmed. Dyspnea (75.0%) was the most common symptom and hypertension (71.1%) was the most common comorbidity. The mean Acute Physiology and Chronic Health Evaluation System (APACHE II) score; Glasgow Coma Score (GCS); Sequential Organ Failure Assessment (SOFA) scores on ICU admission were 17.4 ± 8.2, 12.3 ± 3.9 and 5.9 ± 3.4, respectively. 101 patients (78.1%) received low flow oxygen, 48 had high flow oxygen therapy (37.5%) and 59 (46.1%) had invasive mechanical ventilation. 53 patients (41.4%) had vasopressor therapy and 30 (23.4%) patients had renal replacement therapy (RRT) due to acute kidney injury (AKI). Confirmed patients were more tachypneic (p=0.005) and more hypoxemic than probable patients (p<0.001). Acute respiratory distress syndrome (ARDS) and AKI were more common in confirmed patients than probable (both p<0.001). Confirmed patients had higher values of hemoglobin, C- reactive protein, fibrinogen, D-dimer than probables (respectively, p=0.028, 0.006, 0.000, 0.019). The overall mortality was higher in confirmed patients (p=0.209, 52.6% vs 47.4%). Complications are more common among confirmed COVID-19 patients admitted to ICU. The mortality rate of confirmed COVID-19 patients admitted to the ICU was found to be higher than probable patients. Mortality of confirmed cases were higher than prediction of APACHE-II scoring system.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2021 Fatma İrem Yeşiler, Mesher Çapras, Emre Kandemir, Helin Şahintürk, Ender Gedik, Pınar Zeyneloğlu

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2021-11-13
Published 2022-04-01









