MALAT1 inhibits the Wnt/β-catenin signaling pathway in colon cancer cells and affects cell proliferation and apoptosis
DOI:
https://doi.org/10.17305/bjbms.2019.4408Keywords:
Colon cancer, MALAT1, proliferation, apoptosis, Wnt/β-cateninAbstract
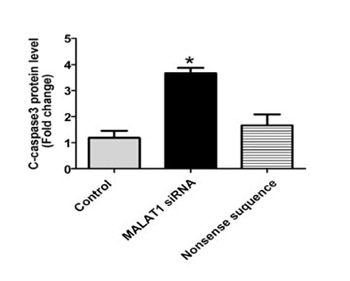
Metastasis associated lung adenocarcinoma transcript 1 (MALAT1) is a highly conserved long noncoding RNA, which has been related to various pathological processes, including cancer. The role and mechanism of MALAT1 in colon cancer are not clear. We investigated MALAT1 expression in colon cancer tissues, the effect of MALAT1 on proliferation and apoptosis of SW480 cells, and the signaling pathway involved in the MALAT1 effects. MALAT1 expression was determined in 60 colon cancer and para-carcinoma tissues using reverse transcription polymerase chain reaction (RT-PCR). Knockdown of MALAT1 in SW480 cells was induced by small interfering RNA (siRNA), and the cells were divided into three groups: untreated control, nonsense siRNA-treated control, and MALAT1 siRNA-treated group. SW480 cell apoptosis was assessed using TUNEL assay and flow cytometry. Apoptosis-related and Wnt/β-catenin signaling pathway-related proteins were detected by Western blotting in SW480 cells. SW480 cell proliferation was assessed by CCK-8 assay. MALAT1 expression was significantly higher in colon cancer vs. para-carcinoma tissues. Knockdown of MALAT1 by siRNA increased the number of apoptotic cells and the apoptosis rate at 24 h post-transfection in SW480 cells. Bcl2 associated X protein (Bax) expression was increased, B-cell lymphoma 2 (Bcl-2) expression was decreased, and the ratio of cleaved caspase-3 to truncated caspase-3 was increased in MALAT1 siRNA-transfected SW480 cells. MALAT1 knockdown decreased the proliferation of SW480 cells at 24 h, 48 h, and 72 h post-transfection. Wnt and β-catenin expression was inhibited in MALAT1 siRNA-transfected SW480 cells. Inhibition of MALAT1 expression in colon cancer may promote apoptosis and hinder cell proliferation by suppressing the activation of Wnt/β-catenin signaling pathway.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2019-10-17
Published 2020-08-03









