Effects of caffeine administration on sedation and respiratory parameters in patients recovering from anesthesia
DOI:
https://doi.org/10.17305/bjbms.2018.2434Keywords:
Caffeine, respiratory insufficiency, anesthesia, sedation, recoveryAbstract
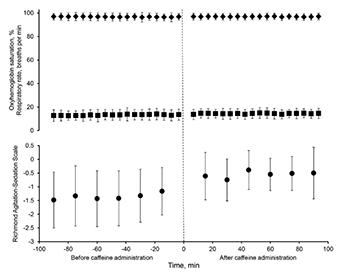
Caffeine has been shown to enhance the speed of recovery from general anesthesia in murine models, though data in human patients is lacking. This is a retrospective review of intravenous caffeine administration (median dose 150 [125, 250] mg) to 151 heavily sedated patients in the post-anesthesia recovery area, to determine the association between caffeine administration and changes in sedation score, respiratory rate, and oxyhemoglobin saturation. Richmond Agitation-Sedation Scale (RASS) score, respiratory rate, and oxyhemoglobin saturation values were obtained during the 90-minute period prior to and following caffeine administration. Generalized estimating equations (GEE) with explanatory variables of time, caffeine, and the time-by-caffeine interaction were created to assess changes in the variables of interest after caffeine administration. Following the administration of caffeine, the RASS scores increased (estimate = 0.57, SE = 0.14, p < 0.001) but a trend over time or in the interaction effect was not observed, suggesting that the changes in RASS were not solely due to the recovery from anesthesia over time. No association was found between caffeine administration and changes in respiratory parameters. No adverse cardiac events were observed. Our data suggests that intravenous caffeine may enhance the speed of recovery following general anesthesia, though future prospective trials are necessary to define the optimal dose and timing of administration.
Citations
Downloads
References
Fredholm BB. Astra award lecture. Adenosine, adenosine receptors and the actions of caffeine. Pharmacol Toxicol 1995;76(2):93-101.
https://doi.org/10.1111/j.1600-0773.1995.tb00111.x.
National Academies Press (US). Caffeine for the Sustainment of Mental Task Performance: Formulations for Military Operations. Washington (DC): National Academies Press (US); 2001.
Ker K, Edwards PJ, Felix LM, Blackhall K, Roberts I. Caffeine for the prevention of injuries and errors in shift workers. Cochrane Database Syst Rev 2010;5:CD008508. https://doi.org/10.1002/14651858.CD008508.
Wang Q, Fong R, Mason P, Fox AP, Xie Z. Caffeine accelerates recovery from general anesthesia. J Neurophysiol 2014;111(6):1331-40. https://doi.org/10.1152/jn.00792.2013.
Weber JG, Klindworth JT, Arnold JJ, Danielson DR, Ereth MH. Prophylactic intravenous administration of caffeine and recovery after ambulatory surgical procedures. Mayo Clin Proc 1997;72(7):621-6.
https://doi.org/10.4065/72.7.621; https://doi.org/10.1016/S0025-6196(11)63567-2.
D’Urzo AD, Jhirad R, Jenne H, Avendano MA, Rubinstein I, D’Costa M, et al. Effect of caffeine on ventilatory responses to hypercapnia, hypoxia, and exercise in humans. J Appl Physiol (1985) 1990;68(1):322-8. DOI: 10.1152/jappl.1990.68.1.322.
Sessler CN, Gosnell MS, Grap MJ, Brophy GM, O’Neal PV, Keane KA, et al. The Richmond Agitation-Sedation Scale: Validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med 2002;166(10):1338-44. https://doi.org/10.1164/rccm.2107138.
Aldrete JA, Kroulik D. A postanesthetic recovery score. Anesth Analg 1970;49(6):924-34. https://doi.org/10.1213/00000539-197011000-00020.
Gali B, Whalen FX, Schroeder DR, Gay PC, Plevak DJ. Identification of patients at risk for postoperative respiratory complications using a preoperative obstructive sleep apnea screening tool and postanesthesia care assessment. Anesthesiology 2009;110(4):869-77. https://doi.org/10.1097/ALN.0b013e31819b5d70.
Rang HP, Dale MM, Ritter JM, Flower RJ. Rang and Dale’s Pharmacology. 6th ed. Amsterdam: Elsevier; 2007.
Lazarus M, Shen HY, Cherasse Y, Qu WM, Huang ZL, Bass CE, et al. Arousal effect of caffeine depends on adenosine A2A receptors in the shell of the nucleus accumbens. J Neurosci 2011;31(27):10067-75. https://doi.org/10.1523/JNEUROSCI.6730-10.2011.
Fong R, Khokhar S, Chowdhury AN, Xie KG, Wong JH, Fox AP, et al. Caffeine accelerates recovery from general anesthesia via multiple pathways. J Neurophysiol 2017;118(3):1591-7. https://doi.org/10.1152/jn.00393.2017.
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2017-11-21
Published 2018-02-20









