Meta-analysis of interleukin 6, 8, and 10 between off-pump and on-pump coronary artery bypass groups
DOI:
https://doi.org/10.17305/bjbms.2017.1505Keywords:
Coronary artery bypass, interleukins, meta-analysis, IL-6, IL-8, IL-10, off-pump coronary artery bypass surgery, on-pump coronary artery bypass surgeryAbstract
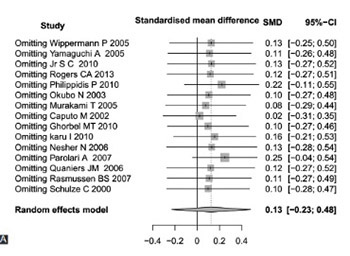
This study aimed to evaluate the role of off-pump coronary artery bypass (CAB) surgery on the decrease of postoperative inflammatory responses in patients. We systematically searched databases of PubMed and Embase to select the related studies. Interleukin (IL) 6, 8, and 10 were used as outcomes and pooled analysis was performed using R 3.12 software. Standardized mean differences (SMDs) and their 95% confidence intervals (95% CIs) were considered as effect estimates. A total of 27 studies, including 1340 participants, were recruited in this meta-analysis. The pooled analyses showed that postoperative concentration of IL-10 at 12 hours was significantly lower in off-pump CAB group compared to on-pump CAB group (SMD = −1.3640, 95% CI = −2.0086-−0.7193). However, no significant differences were found in pre and postoperative concentrations of IL-6 and 8 between off-pump and on-pump CAB groups. These results suggest that there is no advantage of off-pump CAB surgery in the reduction of inflammation compared to on-pump CAB surgery.
Citations
Downloads
References
Brinkman WT, Squiers JJ, Covington KR, Wheeler DA, Arsalan M, Smith RL, et al. Mini-extracorporeal circulation and off-pump techniques associated with less inflammatory gene expression as compared to on-pump in the 24-hour postoperative window following coronary artery bypass grafting. J Cardiothorac Surg 2015;10(Suppl 1):A101. https:/doi.org/10.1186/1749-8090-10-S1-A101.
Afilalo J, Rasti M, Ohayon SM, Shimony A, Eisenberg MJ. Off-pump vs. On-pump coronary artery bypass surgery: An updated meta-analysis and meta-regression of randomized trials. Eur Heart J 2012;33(10):1257-67. https:/doi.org/10.1093/eurheartj/ehr307.
Nesher N, Frolkis I, Vardi M, Sheinberg N, Bakir I, Caselman F, et al. Higher levels of serum cytokines and myocardial tissue markers during on-pump versus off-pump coronary artery bypass surgery. J Card Surg 2006;21(4):395-402.
https:/doi.org/10.1111/j.1540-8191.2006.00272.x.
Wan IY, Arifi AA, Wan S, Yip JH, Sihoe AD, Thung KH, et al. Beating heart revascularization with or without cardiopulmonary bypass: Evaluation of inflammatory response in a prospective randomized study. J Thorac Cardiovasc Surg 2004;127(6):1624-31. https:/doi.org/10.1016/j.jtcvs.2003.10.043.
Ghorbel MT, Cherif M, Mokhtari A, Bruno VD, Caputo M, Angelini GD. Off-pump coronary artery bypass surgery is associated with fewer gene expression changes in the human myocardium in comparison with on-pump surgery. Physiol Genomics 2010;42(1):67-75. https:/doi.org/10.1152/physiolgenomics.00174.2009.
Rogers CA, Pike K, Angelini GD, Reeves BC, Glauber M, Ferrarini M, et al. An open randomized controlled trial of median sternotomy versus anterolateral left thoracotomy on morbidity and health care resource use in patients having off-pump coronary artery bypass surgery: The Sternotomy Versus Thoracotomy (STET) trial. J Thorac Cardiovasc Surg 2013;146(2):306-16.e1-9. DOI: 10.1016/j.jtcvs.2012.04.020.
Brocker C, Thompson D, Matsumoto A, Nebert DW, Vasiliou V. Evolutionary divergence and functions of the human interleukin (IL) gene family. Hum Genomics 2010;5(1):30-55. https:/doi.org/10.1186/1479-7364-5-1-30.
Cua DJ, Sherlock J, Chen Y, Murphy CA, Joyce B, Seymour B, et al. Interleukin-23 rather than interleukin-12 is the critical cytokine for autoimmune inflammation of the brain. Nature 2003;421(6924):744-8. https:/doi.org/10.1038/nature01355.
Kuilman T, Michaloglou C, Vredeveld LC, Douma S, van Doorn R, Desmet CJ, et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell 2008;133(6):1019-31. https:/doi.org/10.1016/j.cell.2008.03.039.
Xing Z, Gauldie J, Cox G, Baumann H, Jordana M, Lei XF, et al. IL-6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest 1998;101(2):311-20. https:/doi.org/10.1172/JCI1368.
Harada A, Sekido N, Akahoshi T, Wada T, Mukaida N, Matsushima K. Essential involvement of interleukin-8 (IL-8) in acute inflammation. J Leukoc Biol 1994;56(5):559-64.
Sato Y, Ohshima T, Kondo T. Regulatory role of endogenous interleukin-10 in cutaneous inflammatory response of murine wound healing. Biochem Biophys Res Commun 1999;265(1):194-9. https:/doi.org/10.1006/bbrc.1999.1455.
Lau J, Ioannidis JP, Schmid CH. Quantitative synthesis in systematic reviews. Ann Intern Med 1997;127(9):820-6. https:/doi.org/10.7326/0003-4819-127-9-199711010-00008.
Andrási TB, Mertens F, Barabás B, Blázovics A. Effect of haemodynamic changes on epithelium-related intestinal injury in off-pump coronary surgery. Heart Lung Circ 2014;23(2):144-51. https:/doi.org/10.1016/j.hlc.2013.07.016.
Ascione R, Lloyd CT, Underwood MJ, Lotto AA, Pitsis AA, Angelini GD. Inflammatory response after coronary revascularization with or without cardiopulmonary bypass. Ann Thorac Surg 2000;69(4):1198-204. https:/doi.org/10.1016/S0003-4975(00)01152-8.
Heijmans JH, Liem KS, Damoiseaux GM, Maessen JG, Roekaerts PM. Pulmonary function and inflammatory markers in patients undergoing coronary revascularisation with or without cardiopulmonary bypass. Anaesthesia 2007;62(12):1233-40.
https:/doi.org/10.1111/j.1365-2044.2007.05254.x.
Johansson-Synnergren M, Nilsson F, Bengtsson A, Jeppsson A, Wiklund L. Off-pump CABG reduces complement activation but does not significantly affect peripheral endothelial function: A prospective randomized study. Scand Cardiovasc J 2004;38(38):53-8. https:/doi.org/10.1080/14017430410024847.
Karu I, Taal G, Zilmer K, Pruunsild C, Starkopf J, Zilmer M. Inflammatory/oxidative stress during the first week after different types of cardiac surgery. Scand Cardiovasc J 2010;44(2):119-24. https:/doi.org/10.3109/14017430903490981.
Caputo M, Yeatman M, Narayan P, Marchetto G, Ascione R, Reeves BC, et al. Effect of off-pump coronary surgery with right ventricular assist device on organ function and inflammatory response: A randomized controlled trial. Ann Thorac Surg 2002;74(6):2088-95. https:/doi.org/10.1016/S0003-4975(02)04025-0.
Massoudy P, Wagner S, Thielmann M, Herold U, Kottenberg-Assenmacher E, Marggraf G, et al. Coronary artery bypass surgery and acute kidney injury – Impact of the off-pump technique. Nephrol Dial Transplant 2008;23(9):2853-60. https:/doi.org/10.1093/ndt/gfn153.
Mazzone A, Gianetti J, Picano E, Bevilacqua S, Zucchelli G, Biagini A, et al. Correlation between inflammatory response and markers of neuronal damage in coronary revascularization with and without cardiopulmonary bypass. Perfusion 2003;18(1):3-8.
DOI: 10.1191/0267659103pf622oa
Murakami T, Iwagaki H, Saito S, Ohtani S, Kuroki K, Kuinose M, et al. Equivalence of the acute cytokine surge and myocardial injury after coronary artery bypass grafting with and without a novel extracorporeal circulation system. J Int Med Res 2005;33(2):133-49. https:/doi.org/10.1177/147323000503300201.
Okubo N, Hatori N, Ochi M, Tanaka S. Comparison of m-RNA expression for inflammatory mediators in leukocytes between on-pump and off-pump coronary artery bypass grafting. Ann Thorac Cardiovasc Surg 2003;9(1):43-9.
Parolari A, Camera M, Alamanni F, Naliato M, Polvani GL, Agrifoglio M, et al. Systemic inflammation after on-pump and off-pump coronary bypass surgery: A one-month follow-up. Ann Thorac Surg 2007;84(3):823-8. https:/doi.org/10.1016/j.athoracsur.2007.04.048.
Philippidis P, Athanasiou T, Nadra I, Ashrafian H, Haskard DO, Landis RC, et al. Anti-inflammatory haemoglobin scavenging monocytes are induced following coronary artery bypass surgery. Eur J Cardiothorac Surg 2010;37(6):1360-6. https:/doi.org/10.1016/j.ejcts.2009.12.043.
Quaniers JM, Leruth J, Albert A, Limet RR, Defraigne JO. Comparison of inflammatory responses after off-pump and on-pump coronary surgery using surface modifying additives circuit. Ann Thorac Surg 2006;81(5):1683-90. https:/doi.org/10.1016/j.athoracsur.2005.11.059.
Rasmussen BS, Laugesen H, Sollid J, Grønlund J, Rees SE, Toft E, et al. Oxygenation and release of inflammatory mediators after off-pump compared with after on-pump coronary artery bypass surgery. Acta Anaesthesiol Scand 2007;51(9):1202-10. https:/doi.org/10.1111/j.1399-6576.2007.01426.x.
Schulze C, Conrad N, Schütz A, Egi K, Reichenspurner H, Reichart B, et al. Reduced expression of systemic proinflammatory cytokines after off-pump versus conventional coronary artery bypass grafting. Thorac Cardiovasc Surg 2000;48(6):364-9. https:/doi.org/10.1055/s-2000-8352.
Serrano CV Jr, Souza JA, Lopes NH, Fernandes JL, Nicolau JC, Blotta MH, et al. Reduced expression of systemic proinflammatory and myocardial biomarkers after off-pump versus on-pump coronary artery bypass surgery: A prospective randomized study. J Crit Care 2010;25(2):305-12. https:/doi.org/10.1016/j.jcrc.2009.06.009.
Al-Ruzzeh S, Hoare G, Marczin N, Asimakopoulos G, George S, Taylor K, et al. Off-pump coronary artery bypass surgery is associated with reduced neutrophil activation as measured by the expression of CD11b: A prospective randomized study. Heart Surg Forum 2003;6(2):89-93. https:/doi.org/10.1532/hsf.1205.
Szerafin T, Brunner M, Horváth A, Szentgyörgyi L, Moser B, Boltz-Nitulescu G, et al. Soluble ST2 protein in cardiac surgery: A possible negative feedback loop to prevent uncontrolled inflammatory reactions. Clin Lab 2005;51(11-12):657-63.
Untch BR, Jeske WP, Schwartz J, Botkin S, Prechel M, Walenga JM, et al. Inflammatory and hemostatic activation in patients undergoing off-pump coronary artery bypass grafting. Clin Appl Thromb Hemost 2008;14(2):141-8. https:/doi.org/10.1177/1076029607308869.
Vallely MP, Bannon PG, Bayfield MS, Hughes CF, Kritharides L. Endothelial activation after coronary artery bypass surgery: Comparison between on-pump and off-pump techniques. Heart Lung Circ 2010;19(8):445-52. https:/doi.org/10.1016/j.hlc.2010.03.006.
Wippermann J, Albes JM, Hartrumpf M, Kaluza M, Vollandt R, Bruhin R, et al. Comparison of minimally invasive closed circuit extracorporeal circulation with conventional cardiopulmonary bypass and with off-pump technique in CABG patients: Selected parameters of coagulation and inflammatory system. Eur J Cardiothorac Surg 2005;28(1):127-32. https:/doi.org/10.1016/j.ejcts.2005.03.032.
Yamaguchi A, Endo H, Kawahito K, Adachi H, Ino T. Off-pump coronary artery bypass grafting attenuates proinflammatory markers. Jpn J Thorac Cardiovasc Surg 2005;53(3):127-32. https:/doi.org/10.1007/s11748-005-0017-7.
Mei YQ, Ji Q, Liu H, Wang X, Feng J, Long C, et al. Study on the relationship of APACHE III and levels of cytokines in patients with systemic inflammatory response syndrome after coronary artery bypass grafting. Biol Pharm Bull 2007;30(3):410-4. https:/doi.org/10.1248/bpb.30.410.
Karu I, Starkopf J, Zilmer K, Zilmer M. Growth factors serum levels in coronary artery disease patients scheduled for bypass surgery: Perioperative dynamics and comparisons with healthy volunteers. Biomed Res Int 2013;2013:985404. https:/doi.org/10.1155/2013/985404.
Shi C, Zhu L, Chen X, Gu N, Chen L, Zhu L, et al. IL-6 and TNF-a induced obesity-related inflammatory response through transcriptional regulation of miR-146b. J Interferon Cytokine Res 2014;34(5):342-8. https:/doi.org/10.1089/jir.2013.0078.
Pickup JC, Mattock MB, Chusney GD, Burt D. NIDDM as a disease of the innate immune system: Association of acute-phase reactants and interleukin-6 with metabolic syndrome X. Diabetologia 1997;40(11):1286-92. https:/doi.org/10.1007/s001250050822.
Phillips CM, Perry IJ. Does inflammation determine metabolic health status in obese and nonobese adults? J Clin Endocrinol Metab 2013;98(10):E1610-9. https:/doi.org/10.1210/jc.2013-2038.
Vozarova B, Weyer C, Lindsay RS, Pratley RE, Bogardus C, Tataranni PA. High white blood cell count is associated with a worsening of insulin sensitivity and predicts the development of Type 2 diabetes. Diabetes 2002;51(2):455-61. https:/doi.org/10.2337/diabetes.51.2.455.
Downloads
Additional Files
Published
How to Cite
Accepted 2016-10-07
Published 2017-05-20









