ERBB4 as a therapeutic target in aortic dissection: Implications for cell-based therapies in vascular regeneration
DOI:
https://doi.org/10.17305/bb.2025.11925Keywords:
Aortic dissection, AD, Erb-B2 receptor tyrosine kinase 4, ERBB4, vascular smooth muscle cells, VSMCs, cell-based therapy, NF-κB pathwayAbstract
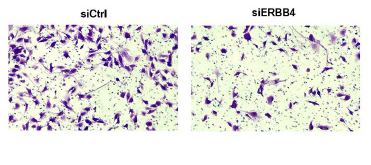
This study investigates the role of Erb-B2 receptor tyrosine kinase 4 (ERBB4) in aortic dissection (AD) pathogenesis and its potential as a therapeutic target in cell-based therapies. Using immunohistochemical (IHC) staining, we examined the phenotypic transition of vascular smooth muscle cells (VSMCs) in thoracic aortic tissue from AD patients. RNA sequencing identified differentially expressed genes (DEGs), highlighting ERBB4 as a key regulator. In vitro, ERBB4 knockdown in human aortic smooth muscle cells (HASMCs) was assessed using cell counting kit-8 (CCK-8 assays), wound healing, transwell migration, colony formation, flow cytometry, and tube formation assays. Our results showed significant inhibition of cell viability, migration, proliferation, and angiogenesis. In vivo, ERBB4 knockdown reduced inflammatory cell infiltration and enhanced collagen fiber contractility, as demonstrated by Masson staining. Mechanistically, ERBB4 silencing suppressed integrin-binding proteins (CD151, ITGAE, ITGB5) and inhibited NF-κB signaling (p-IκBα, p-NF-κB-65). These findings suggest that ERBB4 is critical in AD progression and that its knockdown mitigates pathological changes in HASMCs, reduces inflammation, and restores collagen contractility. ERBB4 may serve as a promising target for cell-based therapies aimed at restoring vascular integrity and function, providing potential new treatment strategies for AD patients.
Citations
Downloads
Downloads
Published
Issue
Section
License
Copyright (c) 2025 Yu Shi, Jinjin Meng, Shengqiang Zhang, Shaofeng Yang, Ge Liu, Zhen Wu, Chongwen Shen, Chao Shi

This work is licensed under a Creative Commons Attribution 4.0 International License.









