Multi-omics reveals that ST6GAL1 promotes colorectal cancer progression through LGALS3BP sialylation
DOI:
https://doi.org/10.17305/bb.2025.11663Keywords:
Colorectal cancer, ST6 β-galactoside α2,6-sialyltransferase, lectin galactoside-binding soluble 3-binding protein, sialylationAbstract
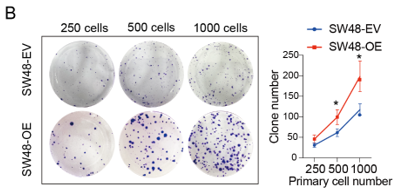
ST6 β-galactoside α2,6-sialyltransferase 1 (ST6GAL1), a crucial enzyme for tumor-associated sialic acid modification, has been reported to positively correlate with colorectal cancer (CRC) tumorigenesis; however, the underlying mechanism remains unclear. To elucidate the protumor mechanisms of ST6GAL1, we performed transcriptomic and N-glycoproteomic analyses and in vitro assays. We found that ST6GAL1 was significantly upregulated in tumor samples than in matched normal samples by analyzing fresh clinical samples from public databases (mean mRNA expression level: tumor vs. normal samples = 0.002712:0.000966, P < 0.05, n = 22). The in vitro results revealed that ST6GAL1 overexpression promoted CRC cell proliferation, migration, and chemoresistance, which were significantly blocked by its knockdown. Transcriptomic data showed that many genes related to the four modules (proliferation/cell cycle, migration, motility, and epithelial–mesenchymal transition (EMT) were upregulated after ST6GAL1 overexpression but downregulated after ST6GAL1 knockdown. Furthermore, the N-glycoproteomic data revealed that 25 substrates that were sialylated upon ST6GAL1 overexpression were related to protumor activity. Importantly, we found that knockdown of lectin galactoside-binding soluble 3-binding protein (LGALS3BP), a newly identified secreted substrate of ST6GAL1, significantly blocked the proliferation, invasion, and chemoresistance of CRC cells induced by ST6GAL1 overexpression. Treatment with sialidases (neuraminidases, NAs) also blocked the protumor activity of ST6GAL1. Thus, ST6GAL1-induced increased sialylation of substrates, such as LGALS3BP and upregulation of protumor genes promote CRC tumorigenesis and chemoresistance, which provides important perspectives and new targets for the treatment of CRC.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2025 Yuanchao Shi, Zhenzhong Pan, Jingwei Duan, Zexing Wang, Yiliang Fang, Bo Tang, Quanlin Guan

This work is licensed under a Creative Commons Attribution 4.0 International License.









