Effect of clozapine on locomotor activity and anxiety-related behavior in the neonatal mice administered MK-801
DOI:
https://doi.org/10.17305/bjbms.2015.472Keywords:
MK-801, Clozapine, Open Field, Elevated Plus Maze, Neonatal miceAbstract
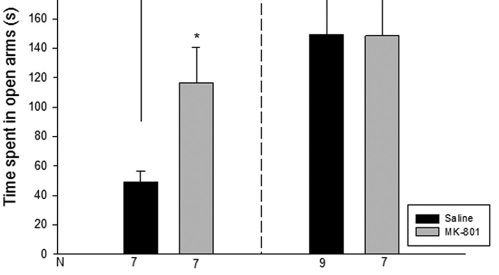
Atypical antipsychotics have been used to treat fear and anxiety disturbance that are highly common in schizophrenic patients. It is suggested that disruptions of N-methyl-d-aspartate (NMDA)-mediated transmission of glutamate may underlie the pathophysiology of schizophrenia. The present study was conducted to analyze the effectiveness of clozapine on the anxiety-related behavior and locomotor function of the adult brain, which had previously undergone NMDA receptor blockade during a developmental period. In order to block the NMDA receptor, male mice were administered 0.25 mg/kg of MK-801 on days 7 to 10 postnatal. In adulthood, they were administered intraperitoneally 0.5 mg/kg of clozapine and tested with open-field and elevated plus maze test, to assess their emotional behavior and locomotor activity. In the group receiving MK-801 in the early developmental period the elevated plus maze test revealed a reduction in the anxiety-related behavior (p<0.05), while the open-field test indicated a decrease in locomotor activity (p<0.01). Despite these reductions, clozapine could not reverse the NMDA receptor blockade. Also, as an atypical antipsychotic agent, clozapine could not reverse impairment in the locomotor activity and anxiety-related behavior, induced by administration of the MK-801 in neonatal period.
Citations
Downloads
References
Viberg H, Ponten E, Eriksson P, Gordh T, Fredriksson A. Neonatal ketamine exposure results in changes in biochemical substrates of neuronal growth and synaptogenesis, and alters adult behavior irreversibly. Toxicology. 2008;249:153-9.
http://dx.doi.org/10.1016/j.tox.2008.04.019
Andersen SL. Trajectories of brain development: point of vulnerability or window of opportunity? Neurosci Biobehav Rev. 2003;27:3-18.
http://dx.doi.org/10.1016/S0149-7634(03)00005-8
Brooks WJ, Weeks AC, Leboutillier JC, Petit TL. Altered NMDA sensitivity and learning following chronic developmental NMDA antagonism. Physiol Behav. 1997;62:955-62.
http://dx.doi.org/10.1016/S0031-9384(97)00169-8
Fredriksson A, Archer T, Alm H, Gordh T, Eriksson P. Neurofunctional deficits and potentiated apoptosis by neonatal NMDA antagonist administration. Behav Brain Res. 2004;153:367-76.
http://dx.doi.org/10.1016/j.bbr.2003.12.026
Guo C, Yang Y, Su Y, Si T. Postnatal BDNF expression profiles in prefrontal cortex and hippocampus of a rat schizophrenia model induced by MK-801 administration. J Biomed Biotechnol. 2010;2010:783297.
http://dx.doi.org/10.1155/2010/783297
Melik E, Babar E, Kocahan S, Guven M, Akillioglu K. Enriched environment has limited capacity for the correction of hippocampal memory-dependent schizoid behaviors in rats with early postnatal NMDAR dysfunction. Int J Dev Neurosci. 2014;33:22-8.
http://dx.doi.org/10.1016/j.ijdevneu.2013.10.004
Scorza MC, Castane A, Bortolozzi A, Artigas F. Clozapine does not require 5-HT1A receptors to block the locomotor hyperactivity induced by MK-801 Clz and MK-801 in KO1A mice. Neuropharmacology. 2010;59:112-20.
http://dx.doi.org/10.1016/j.neuropharm.2010.04.012
Mutlu O, Ulak G, Celikyurt IK, Akar FY, Erden F. Effects of olanzapine, sertindole and clozapine on learning and memory in the Morris water maze test in naive and MK-801-treated mice. Pharmacol Biochem Behav. 2011;98:398-404.
http://dx.doi.org/10.1016/j.pbb.2011.02.009
Mead A, Li M, Kapur S. Clozapine and olanzapine exhibit an intrinsic anxiolytic property in two conditioned fear paradigms: contrast with haloperidol and chlordiazepoxide. Pharmacol Biochem Behav. 2008;90:551-62.
http://dx.doi.org/10.1016/j.pbb.2008.04.014
Ertugrul A, Ozdemir H, Vural A, Dalkara T, Meltzer HY, Saka E. The influence of N-desmethylclozapine and clozapine on recognition memory and BDNF expression in hippocampus. Brain Res Bull. 2011;84:144-50.
http://dx.doi.org/10.1016/j.brainresbull.2010.11.014
Wiley JL, Compton AD, Porter JH. Effects of four antipsychotics on punished responding in rats. Pharmacol Biochem Behav. 1993;45:263-7.
http://dx.doi.org/10.1016/0091-3057(93)90237-N
Millan MJ, Schreiber R, Monneyron S, Denorme B, Melon C, Queriaux S, et al. S-16924, a novel, potential antipsychotic with marked serotonin1A agonist properties. IV. A drug discrimination comparison with clozapine. J Pharmacol Exp Ther. 1999;289:427-36.
Prut L, Belzung C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. European journal of pharmacology. 2003;463:3-33.
http://dx.doi.org/10.1016/S0014-2999(03)01272-X
Sousa N, Almeida OF, Wotjak CT. A hitchhiker's guide to behavioral analysis in laboratory rodents. Genes, brain, and behavior. 2006;5 Suppl 2:5-24.
http://dx.doi.org/10.1111/j.1601-183X.2006.00228.x
Korte SM, De Boer SF. A robust animal model of state anxiety: fear-potentiated behaviour in the elevated plus-maze. European journal of pharmacology. 2003;463:163-75.
http://dx.doi.org/10.1016/S0014-2999(03)01279-2
Pellow S, Chopin P, File SE, Briley M. Validation of open:closed arm entries in an elevated plus-maze as a measure of anxiety in the rat. Journal of neuroscience methods. 1985;14:149-67.
http://dx.doi.org/10.1016/0165-0270(85)90031-7
Cole BJ, Hillmann M, Seidelmann D, Klewer M, Jones GH. Effects of benzodiazepine receptor partial inverse agonists in the elevated plus maze test of anxiety in the rat. Psychopharmacology. 1995;121:118-26.
http://dx.doi.org/10.1007/BF02245598
Martinez G, Ropero C, Funes A, Flores E, Blotta C, Landa AI, et al. Effects of selective NMDA and non-NMDA blockade in the nucleus accumbens on the plus-maze test. Physiology & behavior. 2002;76:219-24.
http://dx.doi.org/10.1016/S0031-9384(02)00704-7
Harris LW, Sharp T, Gartlon J, Jones DN, Harrison PJ. Long-term behavioural, molecular and morphological effects of neonatal NMDA receptor antagonism. The European journal of neuroscience. 2003;18:1706-10.
http://dx.doi.org/10.1046/j.1460-9568.2003.02902.x
Jacobs PS, Taylor BM, Bardgett ME. Maturation of locomotor and Fos responses to the NMDA antagonists, PCP and MK-801. Brain research Developmental brain research. 2000;122:91-5.
http://dx.doi.org/10.1016/S0165-3806(00)00059-6
Akillioglu K, Binokay S, Kocahan S. The effect of neonatal N-methyl-D-aspartate receptor blockade on exploratory and anxiety-like behaviors in adult BALB/c and C57BL/6 mice. Behavioural brain research. 2012;233:157-61.
http://dx.doi.org/10.1016/j.bbr.2012.04.041
Akillioglu K, Babar Melik E, Melik E, Kocahan S. The investigation of neonatal MK-801 administration and physical environmental enrichment on emotional and cognitive functions in adult Balb/c mice. Pharmacol Biochem Behav. 2012;102:407-14.
http://dx.doi.org/10.1016/j.pbb.2012.06.006
Lindefors N, Barati S, O'Connor WT. Differential effects of single and repeated ketamine administration on dopamine, serotonin and GABA transmission in rat medial prefrontal cortex. Brain Res. 1997;759:205-12.
http://dx.doi.org/10.1016/S0006-8993(97)00255-2
Hondo H, Yonezawa Y, Nakahara T, Nakamura K, Hirano M, Uchimura H, et al. Effect of phencyclidine on dopamine release in the rat prefrontal cortex; an in vivo microdialysis study. Brain Res. 1994;633:337-42.
http://dx.doi.org/10.1016/0006-8993(94)91558-X
Lim AL, Taylor DA, Malone DT. A two-hit model: behavioural investigation of the effect of combined neonatal MK-801 administration and isolation rearing in the rat. J Psychopharmacol. 2012;26:1252-64.
http://dx.doi.org/10.1177/0269881111430751
Baier PC, Blume A, Koch J, Marx A, Fritzer G, Aldenhoff JB, et al. Early postnatal depletion of NMDA receptor development affects behaviour and NMDA receptor expression until later adulthood in rats--a possible model for schizophrenia. Behav Brain Res. 2009;205:96-101.
http://dx.doi.org/10.1016/j.bbr.2009.06.018
Amani M, Samadi H, Doosti MH, Azarfarin M, Bakhtiari A, Majidi-Zolbanin N, et al. Neonatal NMDA receptor blockade alters anxiety- and depression-related behaviors in a sex-dependent manner in mice. Neuropharmacology. 2013;73:87-97.
http://dx.doi.org/10.1016/j.neuropharm.2013.04.056
du Bois TM, Huang XF, Deng C. Perinatal administration of PCP alters adult behaviour in female Sprague-Dawley rats. Behav Brain Res. 2008;188:416-9.
http://dx.doi.org/10.1016/j.bbr.2007.11.017
Mc Fie S, Sterley TL, Howells FM, Russell VA. Clozapine decreases exploratory activity and increases anxiety-like behaviour in the Wistar-Kyoto rat but not the spontaneously hypertensive rat model of attention-deficit/hyperactivity disorder. Brain Res. 2012;1467:91-103.
http://dx.doi.org/10.1016/j.brainres.2012.05.047
Lim AL, Taylor DA, Malone DT. Consequences of early life MK-801 administration: long-term behavioural effects and relevance to schizophrenia research. Behav Brain Res. 2012;227:276-86.
http://dx.doi.org/10.1016/j.bbr.2011.10.052
du Bois TM, Deng C, Han M, Newell KA, Huang XF. Excitatory and inhibitory neurotransmission is chronically altered following perinatal NMDA receptor blockade. Eur Neuropsychopharmacol. 2009;19:256-65.
http://dx.doi.org/10.1016/j.euroneuro.2008.12.002
Missale C, Nash SR, Robinson SW, Jaber M, Caron MG. Dopamine receptors: from structure to function. Physiol Rev. 1998;78:189-225.
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2015 Bosnian Journal of Basic Medical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2015-05-19
Published 2015-08-11









