Upregulation of miR-210 promotes differentiation of mesenchymal stem cells (MSCs) into osteoblasts
DOI:
https://doi.org/10.17305/bjbms.2018.2633Keywords:
microRNAs, miR-210, MSC, osteoblast, cord blood, cell differentiationAbstract
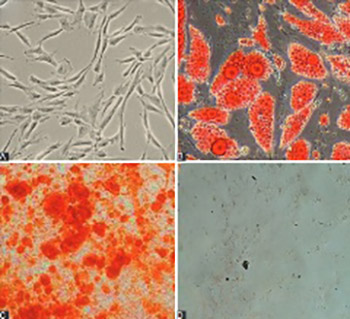
Numerous studies indicated that microRNAs are critical in the regulation of cellular differentiation, by controlling the expression of underlying genes. The aim of this study was to investigate the effect of miR-210 upregulation on differentiation of human umbilical cord blood (HUCB)-derived mesenchymal stem cells (MSCs) into osteoblasts. MSCs were isolated from HUCB and confirmed by their adipogenic/osteogenic differentiation and flow cytometric analysis of surface markers. Pre-miR-210 was amplified from human DNA, digested and ligated with plenti-III-mir-green fluorescent protein (GFP) vector, and cloned in STBL4 bacteria. After confirmation with polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP), the plenti-III-GFP segment bearing pre-miR-210 was transfected into MSCs by electroporation. Two control vectors, pmaxGFP and Scramble, were transfected separately into MSCs. The expression of miR-210 and genes related to osteoblast differentiation, i.e., runt-related transcription factor 2 (Runx2), alkaline phosphatase (ALP) and osteocalcin gene, in the three groups of transfected MSCs was analyzed 0, 7, 14, and 21 days of transfection by quantitative reverse transcription PCR (qRT-PCR). Overexpression of miR-210 was observed in MSCs transfected with miR-210-bearing plasmid, and this was significantly different compared to Scramble group (p < 0.05). Significantly increased expression of Runx2 (at day 7 and 14), ALP and osteocalcin genes (at all time points for both genes) was observed in MSCs with miR-210-bearing plasmid compared to controls. Overall, the overexpression of miR-210 in MSCs led to MSC differentiation into osteoblasts, most probably by upregulating the Runx2, ALP, and osteocalcin genes at different stages of cell differentiation. Our study confirms the potential of miRNAs in developing novel therapeutic strategies that could target regulatory mechanisms of cellular differentiation in various disease states.
Citations
Downloads
References
Khori V, Shalamzari SA, Isanejad A, Alizadeh AM, Alizadeh S, Khodayari S, et al. Effects of exercise training together with tamoxifen in reducing mammary tumor burden in mice: Possible underlying pathway of miR-21. Eur J Phamacol 2015;765:179-87. https://doi.org/10.1016/j.ejphar.2015.08.031.
Kouhkan F, Hafizi M, Mobarra N, Mossahebi-Mohammadi M, Mohammadi S, Behmanesh M, et al. miRNAs: A new method for erythroid differentiation of hematopoietic stem cells without the presence of growth factors. Appl Biochem Biotechnol 2014;172(4):2055-69. https://doi.org/10.1007/s12010-013-0633-0.
Minayi N, Alizadeh S, Dargahi H, Soleimani M, Khatib ZK, Tayebi B, et al. The effect of miR-210 up-regulation on proliferation and survival of mouse bone marrow derived mesenchymal stem cell. Int J Hematol Oncol Stem Cell Res 2014;8(1):15-23.
Chan YC, Banerjee J, Choi SY, Sen CK. miR‐210: The master hypoxamir. Microcirculation 2012;19(3):215-23. https://doi.org/10.1111/j.1549-8719.2011.00154.x.
Mizuno Y, Tokuzawa Y, Ninomiya Y, Yagi K, Yatsuka-Kanesaki Y, Suda T, et al. miR-210 promotes osteoblastic differentiation through inhibition of AcvR1b. FEBS Lett 2009;583(13):2263-8. https://doi.org/10.1016/j.febslet.2009.06.006.
Kosaka N, Sugiura K, Yamamoto Y, Yoshioka Y, Miyazaki H, Komatsu N, et al. Identification of erythropoietin‐induced microRNAs in haematopoietic cells during erythroid differentiation. Br J Hematol 2008;142(2):293-300.
https://doi.org/10.1111/j.1365-2141.2008.07151.x.
Bianchi N, Zuccato C, Lampronti I, Borgatti M, Gambari R. Expression of miR-210 during erythroid differentiation and induction of gamma-globin gene expression. BMB Rep 2009;42(8):493-9. https://doi.org/10.5483/BMBRep.2009.42.8.493.
Huang X, Le QT, Giaccia AJ. MiR-210 - Micromanager of the hypoxia pathway. Trends Mol Med 2010;16(5):230-7. https://doi.org/10.1016/j.molmed.2010.03.004.
Anderson DJ, Gage FH, Weissman IL. Can stem cells cross lineage boundaries? Nat Med 2001;7(4):393-5. https://doi.org/10.1038/86439.
Mohammadi Z, Afshari JT, Keramati MR, Alamdari DH, Ganjibakhsh M, Zarmehri AM, et al. Differentiation of adipocytes and osteocytes from human adipose and placental mesenchymal stem cells. Iran J Basic Med Sci 2015;18(3):259-66.
Bruder SP, Jaiswal N, Haynesworth SE. Growth kinetics, self-renewal, and the osteogenic potential of purified human mesenchymal stem cells during extensive subcultivation and following cryopreservation. J Cell Biochem 1997;64(2):278-94. https://doi.org/10.1002/(SICI)1097-4644(199702)64:2<278::AID-JCB11>3.0.CO;2-F.
Vimalraj S, Selvamurugan N. MicroRNAs: Synthesis, gene regulation and osteoblastdifferentiation. Curr Issues Mol Biol 2013;15(1):7-18.
Williams AR, Hare JM. Mesenchymal stem cells: Biology, pathophysiology, translational findings, and therapeutic implications for cardiac disease. Circulation research 2011;109(8):923-40. http://doi.org/10.1161/CIRCRESAHA.111.243147.
Qiu Y, Chen Y, Zeng T, Guo W, Zhou W, Yang X. EGCG ameliorates the hypoxia-induced apoptosis and osteogenic differentiation reduction of mesenchymal stem cells via upregulating miR-210. Mol Biol Rep 2016;43(3):183-93. https://doi.org/10.1007/s11033-015-3936-0.
Liu XD, Cai F, Liu L, Zhang Y, Yang AL. microRNA-210 is involved in the regulation of postmenopausal osteoporosis through promotion of VEGF expression and osteoblast differentiation. Biol Chem 2015;396(4):339-47. https://doi.org/10.1515/hsz-2014-0268.
Hu R, Liu W, Li H, Yang L, Chen C, Xia ZY, et al. A Runx2/miR-3960/miR-2861 regulatory feedback loop during mouse osteoblast differentiation. J Biol Chem 2011;286(14):12328-39. https://doi.org/10.1074/jbc.M110.176099.
Gong Y, Xu F, Zhang L, Qian Y, Chen J, Huang H, et al. MicroRNA expression signature for Satb2-induced osteogenic differentiation in bone marrow stromal cells. Mol Cell Biochem 2014;387(1-2):227-39. https://doi.org/10.1007/s11010-013-1888-z.
Bhushan R, Grünhagen J, Becker J, Robinson PN, Ott CE, Knaus P. miR-181a promotes osteoblastic differentiation through repression of TGF-β signaling molecules. Int J Biochem Cell Biol 2013;45(3):696-705. https://doi.org/10.1016/j.biocel.2012.12.008.
Downloads
Additional Files
Published
How to Cite
Accepted 2018-01-23
Published 2018-11-07









